Ozempic and Tendon Rupture: Should You Be Worried?
A new study links Ozempic and GLP-1 drugs to higher tendon rupture risk. But the headlines are missing crucial context. A sports medicine physician looks at the data from a different angle.

A patient reaches forward to steady themselves on a handrail. Their pectoralis major/chest muscle tendon suddenly tears. No heavy lifting, no sport, no sudden trauma, just an ordinary movement on an ordinary day. Their doctor has a theory about why. They've been seeing more of this lately, always in patients on the same class of medication: GLP-1 receptor agonists (GLP-1s), the drugs behind household names like Ozempic and Wegovy.
It is a striking clinical pattern. And earlier this month, at one of the world's largest orthopaedic surgery conferences, a research team put some numbers behind it. The results made headlines. But as is so often the case in medicine, the headline is only the beginning of the story.
"The headline is only the beginning of the story."
What the Study Found
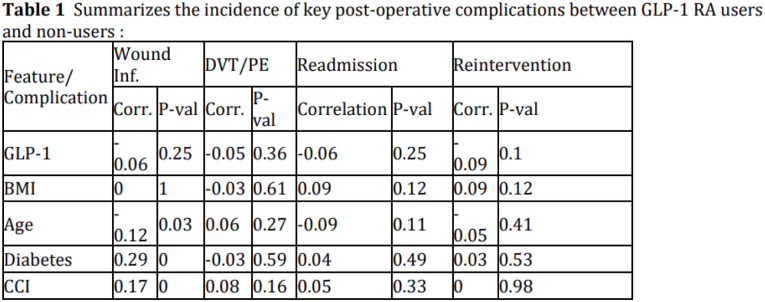
The research, presented at the American Academy of Orthopaedic Surgeons Annual Meeting by a team from the University of Texas, drew on electronic health records from over 70 healthcare organisations. It matched roughly 78,590 people taking GLP-1s with a similar number of people who were not. All of them obese, all with type 2 diabetes.
The finding: GLP-1 users were 1.3 to 1.5 times more likely to rupture a tendon. Five sites were affected: the rotator cuff in the shoulder, the Achilles tendon, the peroneal tendons around the ankle, the pectoralis major/chest muscle tendon, and the quadriceps tendon just above the knee cap.
Before alarm sets in, the authors were clear on one thing: the absolute risk for most of these injuries remained low, under 1% across the board.
The researchers offered three possible explanations for what they were seeing:
- The first is nutritional. Rapid weight loss, alongside the drug's direct effects on tendon cells, may affect the quality of connective tissue, making it more fragile.
- The second is a timing mismatch. Muscle can waste away faster than tendons are able to adapt. When that happens, the tendon ends up absorbing forces it wasn't prepared for.
- The third is what the authors call the activity paradox. Patients who lose significant weight feel better. They move more. They take on physical challenges they hadn't attempted in years. Sometimes their tendons aren't ready for it.
All three are plausible. But they may not be the whole story, or even the most important part of it.
The Question the Study Doesn't Answer
Here is something the headlines did not make room for: people with obesity and type 2 diabetes are already at significantly elevated risk of tendon injury. This is even before they take a single dose of Ozempic or any other GLP-1 drug.
Understanding why requires a brief detour into what high blood sugar actually does to the body over time.
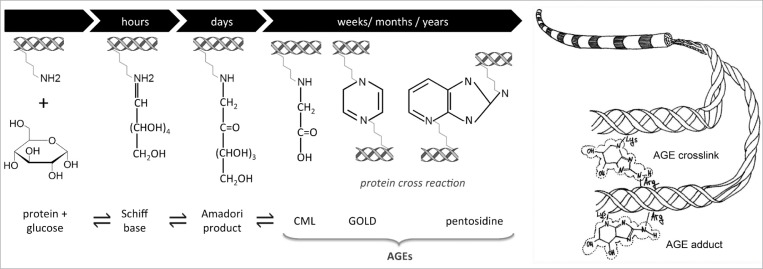
When glucose circulates at high levels, it reacts chemically with proteins like collagen, the main structural material of tendons. The result is the formation of compounds called Advanced Glycation End-products, or AGEs. Think of it as a kind of biological caramelisation. The tendon fibres become stiff, brittle, and less able to absorb the stresses of everyday movement.

AGEs also drive inflammation within the tendon itself, impairing the ability of tendon cells to carry out the routine repairs that keep tissue healthy. The cumulative effect is substantial:
- People with diabetes are three times more likely to develop a tendon disorder. And that's before any weight loss drug enters the picture
- Diabetics face roughly a 44% higher chance of being hospitalised for tendon rupture
- Even being pre-diabetic (an HbA1c above 5.7% but less than 6.5%) is associated with three times the odds of lower limb tendon injury
- Obese individuals have a 4.5 times higher incidence of Achilles tendon rupture compared with those at a healthy weight
These are large numbers. They matter enormously for how we interpret what comes next.
The Problem Baked Into the Data
There is a well-known trap in medical research.
Imagine trying to study whether ambulances cause death. If you simply compared death rates between people who travelled to hospital by ambulance and those who didn't, ambulances would look catastrophically dangerous. The ambulance is not the problem, the underlying emergency is. People who call ambulances are already in crisis. Researchers call this confounding by indication, also known as the sickest patient bias: when sicker patients are more likely to receive a treatment, that treatment can appear to cause the very outcome the illness was already driving.
"When sicker patients are more likely to receive a treatment, that treatment can appear to cause the very outcome the illness was already driving."
Something similar may be at work in this GLP-1 study.
In clinical practice, GLP-1 receptor agonists have historically been reserved for the most difficult-to-treat cases of type 2 diabetes - patients with high cardiovascular risk, long-standing metabolic disease, and those approaching the need for insulin injections. These are precisely the patients who carry the highest burden of silent, cumulative tendon damage from years of poorly controlled blood sugar.
The comparison group, those not taking GLP-1 drugs, almost certainly had better-controlled disease. Lower glucose levels. Less tendon damage already in progress.
So when the study finds that Ozempic users rupture tendons more often, we have to ask: are we seeing a drug effect, or are we simply seeing the natural history of more advanced disease in the patients who needed the drug most?
The study is methodologically careful, but it does not fully untangle those two things.
A Twist in the Data
If GLP-1 drugs were directly damaging tendons at a tissue level, we would expect to see consistent evidence of harm across all the available science. We don't.
If we start with the activity paradox: a study in Diabetes Care found that patients who received more structured, intensive diabetic care had nine times greater risk of lower-extremity tendon injury compared with those receiving standard care. This is not because structured care was harmful, but because those patients became significantly more active. The tendon damage came from the movement, not the medicine.
Then consider the cellular evidence. A 2026 pre-clinical study found that liraglutide, one of the drugs in the GLP-1 class, actually inhibited inflammatory pathways in tendon cells, promoted tissue regeneration, and helped restore mechanical strength. This was in a rat model, which limits how far we can extrapolate the result, but the direction of effect is the opposite of what the headlines imply.
And in patients undergoing Achilles tendon repair surgery, GLP-1 use was associated with an 85% reduction in wound infection, again suggesting that at a biological level, these drugs may actually support tendon healing rather than undermine it.
The picture is complicated. There may be different mechanisms at play. It is not straightforwardly one of harm.
The Bigger Picture
GLP-1 drugs are genuinely remarkable. Their benefits for weight loss, cardiovascular health, and metabolic control are well-documented and, for many patients, life-changing. But a drug class that reaches this deeply into the body's physiology will inevitably involve trade-offs. The question is not whether to look for them, we absolutely should, but whether we are interpreting what we find with the care it deserves.
The chest muscle tears that first caught clinicians' attention are a case in point. The pattern is real and worth taking seriously. But what it most likely reflects is a deficit in muscle mass and physical conditioning - the consequence of significant, rapid weight loss, and not a direct pharmacological attack on tendon tissue. At least not that we know of, yet.
That distinction has practical consequences. If the problem is de-conditioning, the solution is conditioning. It points toward a different model of care: one where starting Ozempic or a similar GLP-1 drug is not the end of the clinical conversation, but the beginning of a more structured one.
- That means building in progressive loading programmes as patients lose weight.
- It means introducing tendon-specific strengthening exercises early.
- It means asking, as newer dual-action drugs like Tirzepatide become more widely used, whether their musculoskeletal profile looks the same or different.
The broader lesson here is about how we consume medical evidence in real time. Headlines are built to travel. They are not built to carry nuance. When a striking finding lands in the news cycle — Ozempic linked to tendon rupture — the questions worth asking are not always in the first paragraph: Who were the patients? What was the comparison group? What does the rest of the evidence say? What might the data be missing?
Those questions take longer to answer. But they are the ones that actually shape good care.
References